How To Draw Molecules Given A Formula
Affiliate 10. Organic Chemistry
10.1 Condensed Structure and Line Structure
Learning Objectives
By the cease of this department, you volition be able to:
- Interpret condense and line structures.
- Draw the condensed construction of a given Lewis Construction or line structure.
- Draw the line structure of a given Lewis Structure or condense structure.
If you wait ahead in this chapter and in other resource at the fashion organic compounds are drawn, you will see that the figures are somewhat dissimilar from the Lewis structures y'all are used to seeing in your full general chemistry book. In some sources, you will see condensed structuresfor smaller molecules instead of full Lewis structures:
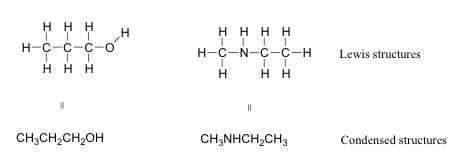
Case 1
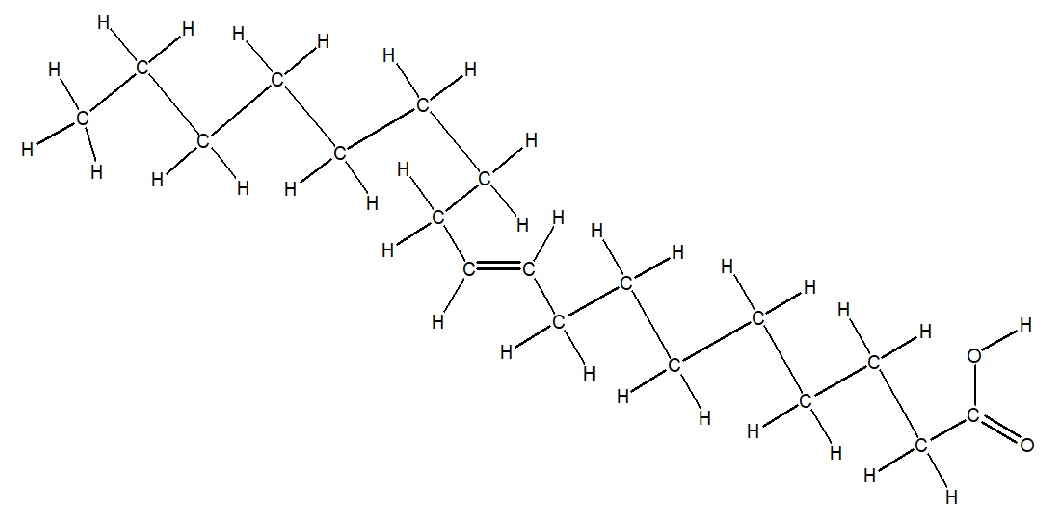
Make up one's mind the Lewis Structure of the following condensed structure of oleic acid, a fatty acid that is institute naturally in various animal and vegetable fats and oils.
CH3(CHii)7CH=CH(CH2)sevenCOOH
Solution
Start by drawing the CH3. The (CHtwo)7 represents a repeating unit, meaning you must describe seven CHtwo'south one subsequently some other, which are bonded to a CH which is bonded to a CH, and then another seven CHtwo's. The COOH represent a carboxylic acid, which ways you have a C=O connected to an O-H. Always double check your structure to ensure every carbon is making four bonds. When you do this, you will come across the 2 CH must be double bonded.
Exam Yourself
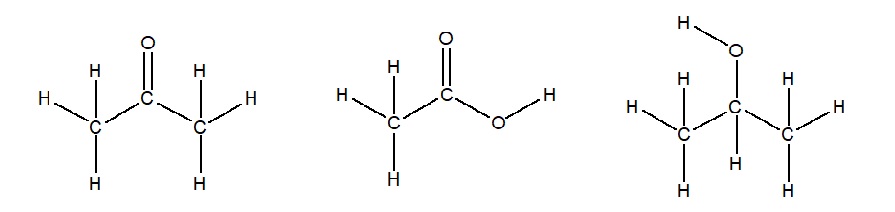
Common organic compounds that you likely have at home are: acetone (CH3COCHthree) found in nail smooth remover, acetic acid (CH3COOH) found in vinegar, and isopropanol ((CHthree)2CHOH) constitute in rubbing alcohol. Decide the Lewis Structure for each of these household chemicals.
Answer
More unremarkably, organic and biological chemists use an abbreviated drawing convention called line structures, also known as skeletal structures or line bond structures. The convention is quite unproblematic and makes it easier to describe molecules, but line structures do take a little fleck of getting used to. Carbon atoms are depicted not past a capital C, simply by a 'corner' between two bonds, or a free end of a bond. Open-concatenation molecules are usually drawn out in a 'zig-zig' shape. Hydrogens attached to carbons are generally not shown: rather, similar alone pairs, they are simply implied (unless a positive formal charge is shown, all carbons are assumed to have a full octet of valence electrons). Hydrogens bonded to nitrogen, oxygen, sulfur, or anything other than carbon areshown, only are usually drawn without showing the bond. The post-obit examples illustrate the convention.
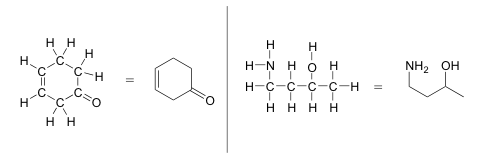
As you can see, the 'pared down' line construction makes it much easier to see the basic structure of the molecule and the locations where there is something other than C-C and C-H single bonds. For larger, more complex biological molecules, it becomes impractical to utilise full Lewis structures. Conversely, very small-scale molecules such as ethane should exist fatigued with their total Lewis or condensed structures.
Sometimes, one or more than carbon atoms in a line structure will exist depicted with a capital C, if doing so makes an explanation easier to follow.If you characterization a carbon with a C, you also must draw in the hydrogens for that carbon.
Instance 2
Draw the line structures for these ii molecules:
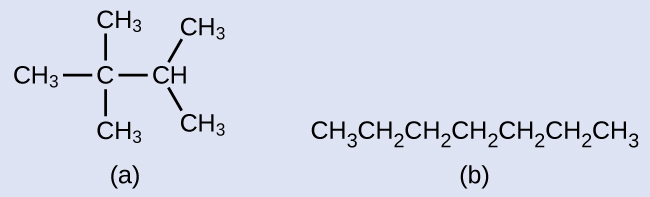
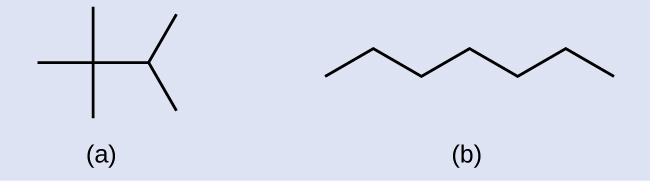
Solution
Each carbon atom is converted into the finish of a line or the place where lines intersect. All hydrogen atoms fastened to the carbon atoms are left out of the structure (although we yet demand to recognize they are at that place):
Test Yourself
Describe the line structures for these ii molecules:
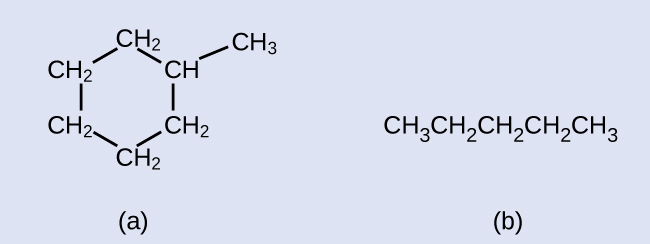
Answers
Example 3
Identify the chemical formula of the molecule represented here:
Solution
At that place are eight places where lines intersect or end, significant that in that location are viii carbon atoms in the molecule. Since nosotros know that carbon atoms tend to make four bonds, each carbon atom will have the number of hydrogen atoms that are required for four bonds. This compound contains 16 hydrogen atoms for a molecular formula of C8Hsixteen.
Location of the hydrogen atoms:
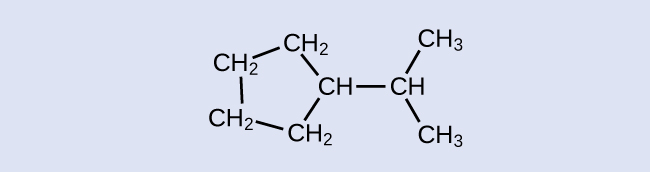
Test Yourself
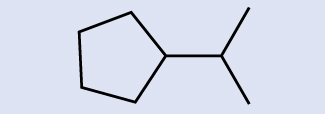
Place the chemical formula of the molecule represented here:
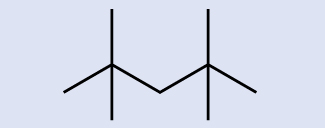
Answer
C9H20
Case iv
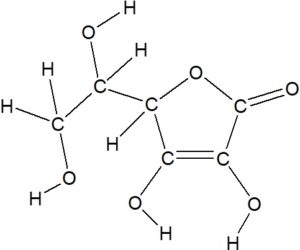
Determine the Lewis Structure of the following line construction of L-ascorbic acid (vitamin C):
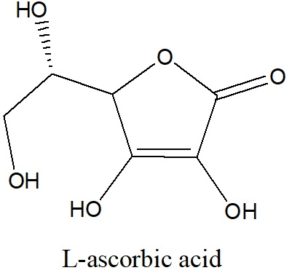
Solution
At each corner or intersection of lines or cease of a line, add a C:
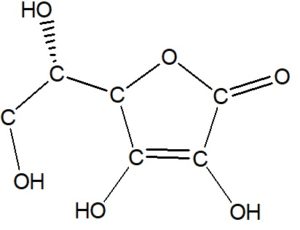
Each C makes fours bonds. If some are non visible, and then add a bond to H for each bonds missing. Also show the bond between the O's and H's.
Test Yourself
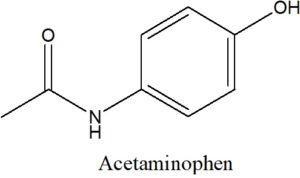
Determine the Lewis Structure of the following line structure of acetaminophen, the pain and fever medicine constitute in Tylenol.
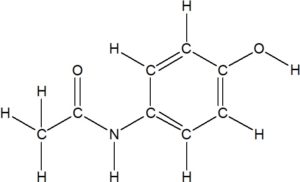
Answer
Key Concepts and Summary
Condensed structures and line structures are a fashion organic structures can be represented in a very concise manner.
Exercises
1. Draw the line bond structure for the following compounds:
a) (HO)3C(CHii)iiN(CH2CHO)CH(CHtwoCHiii)two
b) CH3(CHii)3CH(CH3)(CH2)2OCH2CHiii
c) HOOCCHtwoO(CHtwo)2Northward(CH2CH3)2
d) HOCCH2CH(CH3)CH2CH(CHthree)(CH2)2COOCH3
e) H2NCHtwo[CH(CH3)]2(CH)fourCO(CH2)twoCHiii
f) ClCH2CH(CH3)COCHtwoOCH2CO(CHtwo)twoCH3
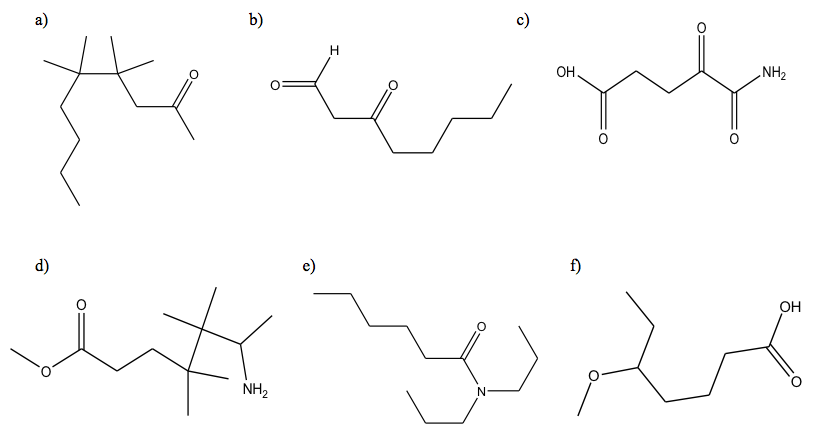
2. For the following compounds,give the chemic formula and the condensed structure:
Answers
one.
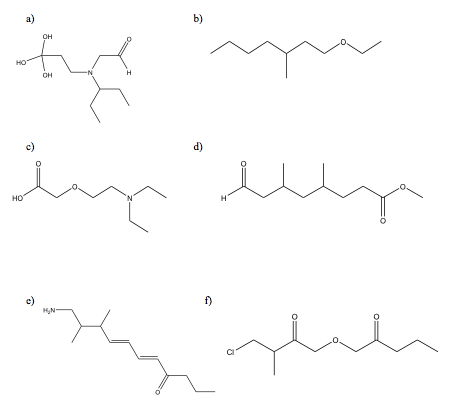
2. a) C13H26O; CH3(CHtwo)3C(CHiii)2C(CH3)2CHiiCOCHiii
b) C8H14Oii; CHOCHiiCO(CHii)fourCH3
c) CfiveH7OivNorthward; COOH(CH2)2(CO)2NH2
d) C12H25O2N; CH3OCO(CHii)2C(CH3)iiC(CH3)2CH(CH3)NH2
e) C12H25ON; CH3(CHii)4CON(CHiiCH2CHthree)2
f) CviiiHsixteenO3; CH3CH2CH(OCH3)(CH2)3COOH
Source: https://pressbooks.bccampus.ca/chem1114langaracollege/chapter/9-1-condensed-structure-and-line-structure-cw/
Posted by: shroyerplasoner.blogspot.com


0 Response to "How To Draw Molecules Given A Formula"
Post a Comment